Bring on the Clones
Conception and misconception
Natural diversity
Creativity is a word that captures the awe of an unfolding universe; it is the mother of novelty. I won’t apologize for what seems to imply a necessary creator, because there has been novelty at every stage of evolution since the Big Bang—from the formation of quarks to living cells to complex brains.
The first cells emerged as prokaryotes that replicated themselves by cloning, but they could not stay uniform under the bombardment of high background radiation and a likely deficit of nucleic acid proofreading. Like conjugating bacteria today, we think that some began pairing up to transfer plasmid DNA to a partner, spreading fitness-enhancing traits. This was a step before the emergence of sexual reproduction, which set eukaryotes on a course toward greater variation for natural selection to pick winners.
The etymology of reproduction is to bring forth again, or to copy, but that fails to represent biology, in which something novel is created in each generation by refreshing the genome. The word appears for the first time in English in the seventeenth century, but early scientists, including William Harvey, spoke only of generation, which doesn’t convey the meaning of copying.
The evolution of sex with two partners (only two) has been so successful that it has persisted ever since. Only rarely have species reverted to asexual copying, and in viviparous mammals, it is prohibited by genomic imprinting. The creative act of sex generates unique offspring by randomly assorting genes in meiosis and the union of genetically different cells. The probability that you or I are identical at every DNA base pair with someone in the past, present, or future is astronomically remote, with a single exception—monozygotic twins, although they too gradually diverge epigenetically over time. The generation of immense variation was necessary for life to flourish on Earth.
The probability of a human embryo splitting into two genetically identical babies is about four in a thousand worldwide (and more often with IVF). Twinning is uncommon, and higher orders are progressively rarer. Annette Dionne died recently, the last of the unique Canadian identical quintuplets. If we assume, simplistically, an equal probability at every split of their original embryo, their chances of birth were one in 3.9 billion. But biology is never “simple”, and the likelihood of similar quintuplets is bound to be much less. Nature seems to hate uniformity, and differences we see at the whole-body level are found in organs, cells, and organelles.
Doppelgängers
The word “cloning” means different things in biology and technology, but reproductive cloning in common parlance implies making an exact copy of a body—a perversion of how nature behaves.
Vegetative propagation in plants is a special case. Horticulturists have practiced grafting from time immemorial because somatic cells in plants can revert to totipotency by dedifferentiating to meristems that can regenerate a whole plant.
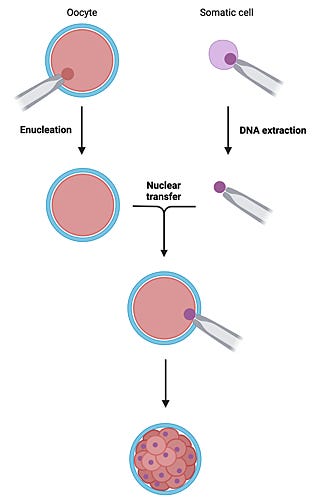
This never happens in vertebrate animals—although genetic information encoded by DNA in the zygote is not lost afterwards. The late John Gurdon made this key discovery by cloning frogs through serial transfer of nuclei from tadpole intestinal cells into enucleated eggs to reprogram the genome for unfolding development to the adult stage. Yet to date, no one has succeeded with adult cells from amphibians.
Gurdon’s student in Oxford, Derek Bromhall, is reported to have adapted the method for rabbits to generate a morula, but without transferring it to a surrogate uterus, we still didn’t know if mammalian clones could survive. However, a few viable sheep and cattle clones were produced by nuclear transfer of embryonic cells and by cloning nature’s way—splitting embryos at the 2- or 4- or 8-cell stage—but low efficiency limited commercial applications for spreading genetically valuable livestock.
A woolly celebrity
Meanwhile, some colleagues were quietly making progress at the Roslin Institute outside Edinburgh, where I worked on fertility preservation. The team leader, Ian Wilmut, inspired the project, Keith Campbell had a key insight about cell-cycle synchronization, and Bill Ritchie—now the sole survivor of the trio—was the embryologist responsible for nuclear transfer and cell culture.
Their work came to widespread attention when they created the lambs Megan and Morag using differentiated cells from early embryos. Mature eggs extracted from donor ewes were enucleated by Bill, who inserted a donor cell under the zona pellucida, then fired an electrical impulse for fusion with the egg membrane. Activation of the reconstructed cell formed a pseudopronucleus. In successful cases, after six or seven cell divisions, the clone formed a blastocyst for transfer to the uterus of a receptive ewe.
I lost touch with the team that kept progress under wraps after I moved south, so I was unprepared when a journalist called my office in 1997. He wanted my reaction to news of a clone born after nuclear transfer from an udder cell stored in the freezer from the biopsy of a deceased adult sheep. The press broke an embargo on early publication imposed by Nature because the temptation to trigger a public sensation was too great to resist. I was flabbergasted. Amid the publicity, cartoons, and questions from politicians and ethicists, Ian’s team was trapped in a frenzy of attention they had neither expected nor wanted. “When will they stop pursuing us?” he asked me several weeks later, looking downcast.
But Dolly—the first clone from an adult mammal—was a big deal, and her legacy resonates decades later. They named her after the famous American singer, but no one has composed a song to celebrate her, so here are my lyrics adapted from a popular song in the 1920s: If You Knew Susie.
If you knew Dolly, like I know Dolly
Oh! Oh! Oh! What a sheep!
There’s nothing more snazzy
Than a ewe made by ovoplasty
Oh! Oh! Holy Moses, what a chassis …
It took 277 attempts at somatic cell nuclear transfer to produce the embryo that became Dolly. Some dismissed the achievement as a fluke; others demanded rigorous tests to validate her; and some journalists even asked if it was fraudulent—so shocked were people across a wide spectrum who had previously believed cloning is impossible.
The skeptics were soon answered by confirmatory studies and new cases. One of the virtues of science is that once a breakthrough is made, no matter how high the starting hurdle, it becomes easier as methods are refined, and then they are adapted for new applications.
Some sixteen mammalian species have been cloned so far, although I might have undercounted some varieties of farm animals, domestic pets, or endangered species. The commercial value of the technology rises as more livestock of superior genetic worth are generated, shortening the generation interval by using donor cells from calves, and using CRISPR gene editing tools to produce bespoke cattle that grow faster, produce more milk, and yield better steaks. For a tidy sum, your dog or cat can be cloned—even post-mortem—but no doppelgänger will ever replace my pets.
After Dolly, the Roslin team created the first two transgenic sheep clones. Polly and Molly made human blood factor IX in their milk for treating hemophilia, an advance on fractionating blood from healthy donors, and before it was produced in bioreactors by recombinant genetic engineering. Animal cloning spurred stem-cell technology for therapeutic cloning, and also inspired the discovery of induced pluripotent stem cells, which are revolutionizing regenerative medicine.
Now for some snags
Dolly was closely observed for signs of ill health until six years old, when she had to be euthanized—the same age as her donor nucleus, rather coincidentally. If the nucleus carried a memory of the age of the donor cell, clones would die young from being “older” by having correspondingly short telomeres. Telomerase extends these chromosomal “caps” in stem cells and cancer cells, and it is also present in egg cytoplasm. Dolly’s telomeres told a complex story, but cloned animals otherwise have lengths more commensurate with their developmental age.
Keith moved to Nottingham, where his team cloned five more healthy sheep. Does that mean all’s well? Not exactly. Cloning is inefficient and generates far more fetal losses and birth defects than normal reproduction, despite recent progress. It should not surprise us if a diploid nucleus implanted into egg cytoplasm is not accurately reprogrammed or if development is slower. The problem is not from corruption of the DNA sequence but dysregulation of gene expression. It is epigenetic. This is not the place to describe a complicated picture, so I take liberties—
The first challenge is to erase the epigenetic memory of the donor nucleus. TET enzymes remove covalent methyl groups at CpG nucleotides in DNA, which silences genes not needed by differentiated cells. The genome turns back to the totipotent state, like a zygote. Remethylation then proceeds to mark future cell lineages on that blank slate. It is easy to imagine errors in a process that short-circuits the normal stages of gametogenesis.
Imprinted genes are special cases that have complementary roles in development. They are expressed by only one of a pair of alleles: genes with a paternal origin are needed for placental growth, while the maternal copies have roles in fetal development. Experiments in mice, for instance, show hypomethylation of the H19/Igf2 locus suppresses fetal growth, whereas hypermethylation of the same causes fetal overgrowth, which mirrors a human overgrowth syndrome. Errors can creep in.
The expression of genes on the X chromosome is also regulated by methylation. One of the pair of X chromosomes in female cells is randomly inactivated after the blastocyst stage by the Xist gene product, so that females, like males, have a single active X. When a somatic nucleus is inserted into the egg’s cytoplasm, the inactive X must be demethylated before remethylation at specific loci for embryonic lineages to differentiate. The methylation status of Xist is crucial to avoid disordered gene expression.
Gene activity is also affected by the cloud of proteins that give structure to chromatin. DNA spools around octuplets of histone proteins in nucleosomes, and their chemically modified tails act as on/off switches affecting the access of transcription factors to gene promoters.
We still can’t grasp the immensity of epigenetic modifications needed for normal development, which clones need to negotiate for thriving and avoiding abnormality. No wonder cloning has a mystique as embryologists transfer a nucleus on a wing and a prayer. Any yet, progress rolls forward, currently focused on long non-coding RNAs despite limited supplies of eggs for research.
The applications and efficiency of reproductive cloning are growing, yet it remains an immature technology, hedged by ethical concerns—not least animal welfare. The fires of debate over human cloning smoldered for two decades after Dolly, then heated up again when macaque monkeys were cloned in 2018. Although banned in many countries, a few zealots still ask: if not now, when will we see the first cloned human baby? I ask if technology—or any technology—can be proscribed absolutely, forever, and everywhere? There were rumors that human clones already exist.
As long ago as 1978, the science writer David Rorvik published In His Image as a factual account of a childless man called Max who wanted to generate an heir by cloning to inherit his fortune. A scientist used Dr. Bromhall’s nuclear transfer technique (when human eggs were as rare as hen’s teeth), and the embryo was implanted in a poor woman called Sparrow to gestate Baby Adam. I remember colleagues avoided discussing the claim, perhaps unsure how to react, when the book burst on the bestseller list and created as much condemnation as public sensation, magnified by the timely birth of Louise Brown. Evidently, the author never denied his story, but I heard that Bromhall sued the publisher after loaning his thesis to the millionaire. What do you make of this bizarre story?
More recently, there have been attempts to clone human embryos by maverick researchers in South Korea and the United States, both of them unsuccessful and poorly documented. They received as much scorching criticism as they deserved. I cannot see any justifiable reason to clone humans, yet it will happen someday.
My skepticism is more than prime concerns over safety and a child’s social welfare. There are at least three misconceptions about cloning: it can’t be an exact copy of the original when the donor cell harbors somatic mutations; nor will the epigenome be identical; nor will the mitochondrial genomes be the same. Nature hates to copy.
Afterthought
When all is said and done, ethics cannot be outsourced to technology or postponed until after the fact. Reproductive cloning tests not just the limits of what we can do, but how deeply we understand human development, respect individuality, and behave responsibly for future lives. Biology teaches us that copying is never exact or neutral. If human cloning does occur, the question will not be whether it was technically possible, but if it was undertaken with humility, restraint, and regard for the child rather than the experiment.
Thanks to Dr. William Ritchie for checking facts and adding details
Image: Somatic cell nuclear transfer technique (BioRender)
The author uses generative AI for copy-editing and images, nothing else


Thank you Roger - as always, clearly explained and thought-provoking.
Am I the only one a little concerned about high-profile multi-billionaires (especially those who believe they are above all ethical considerations) wanting to clone themselves for 'posterity'? Humility, restraint and responsibility wouldn't factor in their plans.
Thank goodness for your list of cloning misconceptions...